Our paper reporting the mechanism of NO reduction by Cu cluster was published in ACS Omega (ACS).
「Catalysis of Cu Cluster for NO Reduction by CO: Theoretical Insight into Reaction Mechanism」
Takagi, Nozomi; Ishimura, Kazuya; Miura, Hiroki; Shishido, Tetsuya; Fukuda, Ryoichi; Ehara, Masahiro; Sakaki, Shigeyoshi
ACS Omegan, 2019, 4(2), pp. 2596- DOI:10.1021/acsomega.8b02890 [Open Access]
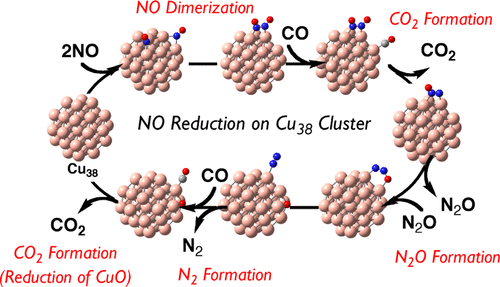
Density functional theory calculations here elucidated that Cu38-catalyzed NO reduction by CO occurred not through NO dissociative adsorption but through NO dimerization. NO is adsorbed to two Cu atoms in a bridging manner. NO adsorption energy is much larger than that of CO. N-O bond cleavage of the adsorbed NO molecule needs a very large activation energy (ΔG°‡). On the other hand, dimerization of two NO molecules occurs on the Cu38 surface with small ΔG°‡ and very negative Gibbs reaction energy (ΔG°) to form ONNO species adsorbed to Cu38. Then, a CO molecule is adsorbed at the neighboring position to the ONNO species and reacts with the ONNO to induce N-O bond cleavage with small ΔG°‡ and very negative ΔG°, leading to the formation of N2O adsorbed on Cu38 and CO2 molecule in the gas phase. N2O dissociates from Cu38, and then it is readsorbed to Cu38 in the most stable adsorption structure. N-O bond cleavage of N2O easily occurs with small ΔG°‡ and significantly negative ΔG° to form the N2 molecule and the O atom adsorbed on Cu38. The O atom reacts with the CO molecule to afford CO2 and regenerate Cu38, which is rate-determining. N2O species was experimentally observed in Cu/γ-Al2O3-catalyzed NO reduction by CO, which is consistent with this reaction mechanism. This mechanism differs from that proposed for the Rh catalyst, which occurs via N-O bond cleavage of the NO molecule. Electronic processes in the NO dimerization and the CO oxidation with the O atom adsorbed to Cu38 are discussed in terms of the charge-transfer interaction with Cu38 and Frontier orbital energy of Cu38.